��Ŀ����
��ͼ��ʾΪ������ļס����������أ��Իش�
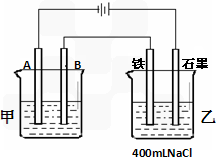
��1�����׳����õ��ԭ�������϶�������A���ĵ缫����Ϊ__________��Ӧѡ�õĵ������Һ��_____________��B���ĵ缫��ӦʽΪ____________��
��2���ҳ���������������̪��Һ����ʼ���һ��ʱ�䣬�缫�����ʺ�ɫ����_______�������������ʯī����
��3�����׳����������仯Ϊ43.2�ˣ����Ҳ��������Ϸų��������ڱ���µ������_______ml�������Ҳ�����Һ����ı仯,�����������Һ�����ʵ���Ũ��Ϊ________, ��pHֵ����___________.��������������ˮ�е��ܽ⣩��
��2���ҳ���������������̪��Һ����ʼ���һ��ʱ�䣬�缫�����ʺ�ɫ����_______�������������ʯī����
��3�����׳����������仯Ϊ43.2�ˣ����Ҳ��������Ϸų��������ڱ���µ������_______ml�������Ҳ�����Һ����ı仯,�����������Һ�����ʵ���Ũ��Ϊ________, ��pHֵ����___________.��������������ˮ�е��ܽ⣩��
��1������AgNO3��Ag -e- = Ag+
��2����
��3��4480mL��1mol/L��14
��2����
��3��4480mL��1mol/L��14

��ϰ��ϵ�д�
�����Ŀ
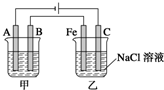 ��ͼ��ʾ���ס���Ϊ������������أ��Իش�
��ͼ��ʾ���ס���Ϊ������������أ��Իش�
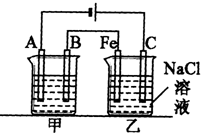
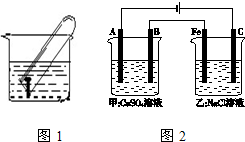 �绯ѧ�ڹ�ҵ�������й㷺Ӧ�ã�
�绯ѧ�ڹ�ҵ�������й㷺Ӧ�ã�