��Ŀ����
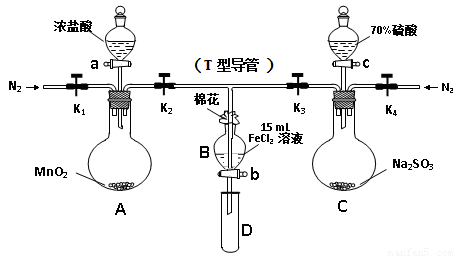
Ϊ��֤������Cl2 > Fe3+ > SO2��ijС������ͼ��ʾװ�ý���ʵ�飨�г�������A�м���װ�����ԣ��������Ѽ��飩��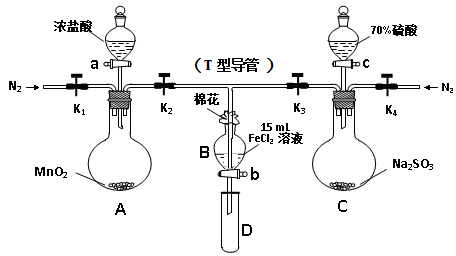
ʵ����̣�
���ɼ�K1~K4��ͨ��һ��ʱ��N2���ٽ�T�͵��ܲ���B�У� ����ͨ��N2��Ȼ��ر�K1��K3��K4��
����a���μ�һ������Ũ���ᣬ����A��
��B����Һ���ʱ��ֹͣ���ȣ��н�K2��
��������b��ʹԼ2mL����Һ����D�Թ��У����������е����ӡ�
������K3�ͻ���c������70%�����ᣬһ��ʱ���н�K3��
���������Թ�D���ظ����̢���
��1�����̢��Ŀ���� ��
��2�����н������ҺΪ ��
��3��A�з�����Ӧ�Ļ�ѧ����ʽΪ ��
��4����˵��������Fe3+ > SO2�����ӷ���ʽ�� ��
��5�����̢�������B��Һ���Ƿ���SO42-�IJ����� ��
��6���ס��ҡ�����λͬѧ�ֱ����������ʵ�飬���ǵļ�������ܹ�֤��������Cl2 > Fe3+ > SO2���� ����ס����ҡ��������������ñ�Ҫ������˵������ ��
| | ���̢� B��Һ�к��е����� | ���̢� B��Һ�к��е����� |
| �� | ��Fe3+��Fe2+ | �� Fe2+ |
| �� | ����Fe3+����Fe2+ | ��SO42- |
| �� | ��Fe3+��Fe2+ | ��SO42- |
�������ϣ� Fe2+(aq)+ SO32- (aq)
 FeSO3(s)��ī��ɫ��������裺FeCl3�� SO2�ķ�Ӧ�������м����FeSO3����Һ�ĺ���ɫ��FeSO3��ī��ɫ����FeCl3����ɫ���Ļ��ɫ��
FeSO3(s)��ī��ɫ��������裺FeCl3�� SO2�ķ�Ӧ�������м����FeSO3����Һ�ĺ���ɫ��FeSO3��ī��ɫ����FeCl3����ɫ���Ļ��ɫ��ijͬѧ�������ʵ�飬֤ʵ�ü��������

�� ��ҺE��F�ֱ�Ϊ �� ��д��ѧʽ����
�����û�ѧƽ��ԭ�����Ͳ���3�к���ɫ��Һ��ɫ��Ϊdz��ɫ��ԭ�� ��
��18�֣���1���ų�װ���еĿ�������ֹ���� ��2�֣� ��2��NaOH��Һ ��2�֣�
��3��MnO2 + 4HCl(Ũ) MnCl2 + Cl2��+ 2H2O ��2�֣�
MnCl2 + Cl2��+ 2H2O ��2�֣�
��4��2Fe3+ + SO2 + 2H2O��2Fe3+ + SO42-+4H+��2�֣�
��5����D�Թ��м����������ᣬ�ټ�BaCl2��Һ�����а�ɫ����������˵����Һ�к���SO42- �������𰸾����֣���2�֣�
��6���� ��2�֣� , �˼����ֻ��֤��������Cl2 > Fe3+ ������֤��Fe3+ > SO2����ΪCl2Ҳ������SO2����SO42- ��2�֣�
��7���� Na2SO3����������������Σ���FeCl3 ��2�֣���1�֣�
�� Fe3+ ���� SO32-��c(SO32-)��С��ʹƽ��Fe2+ (aq)+ SO32- (aq)  FeSO3(s)�����ƶ�����Һ��ɫ�ɺ���ɫ��Ϊdz��ɫ ��2�֣�
FeSO3(s)�����ƶ�����Һ��ɫ�ɺ���ɫ��Ϊdz��ɫ ��2�֣�
���������������1������װ���к��п������������Ĵ��ڣ����ʵ��������ţ����Թ��̢��Ŀ�����ų�װ���еĿ�������ֹ���š�
��2��������SO2���Ǵ�����Ⱦ����������ŷţ�������������ҺӦΪNaOH��Һ����������β����
��3��װ��A���Ʊ������ģ���Ӧ�Ļ�ѧ����ʽ��MnO2 + 4HCl(Ũ) MnCl2 + Cl2��+ 2H2O��
MnCl2 + Cl2��+ 2H2O��
��4�������Ӿ��������ԣ��ܰ�SO2�����������ᣬ��Ӧ�����ӷ���ʽ��2Fe3+ + SO2 + 2H2O��2Fe3+ + SO42-+4H+��
��5������B��Һ���Ƿ���SO42-�IJ�������D�Թ��м����������ᣬ�ټ�BaCl2��Һ�����а�ɫ����������˵����Һ�к���SO42-��
��6�����̢�B��Һ����Fe3+��Fe2+��˵�����������������������������������ǿ�������ӵġ����̢�B��Һ�к����������ӣ�˵��SO2�ܰ������ӻ�ԭ�����������ӣ���˵����������������ǿ��SO2��������ȷ�ģ�ͬ��������Ҳ����ȷ�ģ����ڱ��У������ֻ��֤��������Cl2 > Fe3+ ������֤��Fe3+ > SO2����ΪCl2Ҳ������SO2����SO42-�����Ա��Ǵ���ġ�
��7���� ʵ������Һ��ī��ɫ��˵��ƽ��������Ӧ�����ƶ������Լ����Ӧ����Na2SO3����������������Σ���������2����Һ�Ժ���ɫ��˵�������Ӧ����FeCl3 ��
�� ����Fe3+ ���� SO32-��c(SO32-)��С��ʹƽ��Fe2+ (aq)+ SO32- (aq) FeSO3(s)�����ƶ�����Һ��ɫ�ɺ���ɫ��Ϊdz��ɫ��
FeSO3(s)�����ƶ�����Һ��ɫ�ɺ���ɫ��Ϊdz��ɫ��
���㣺����������ԭ��Ӧ���жϣ�����ʵ��������������Ʊ����������Ȼ�����SO2���ʵ�̽���Լ����������ƽ��״̬��Ӱ��
�����������Ǹ߿��еij������ͣ��Ѷȴ��ۺ���ǿ����ѧ����Ҫ��ߡ�������ע�ضԻ���֪ʶ���̺�ѵ����ͬʱ�����ض�ѧ�������������ͽ��ⷽ����ָ����ѵ��������������ѧ���淶�Ͻ���ʵ����������Լ���������������������Ҫ���Գ���������ѡ�á�ʵ���������Ϊ���ģ�ͨ����ʲô��Ϊʲô���������ص㿼��ʵ����������Ĺ淶�Ժ�ȷ�Լ��������֪ʶ���ʵ����������������������ۺ���ǿ�����ۺ�ʵ������ϵ���ܣ��еĻ��ṩһЩ�µ���Ϣ�����Ҫ��ѧ���������桢ϸ�µ����⣬��ϵ��ѧ����֪ʶ�ͼ��ܣ�����֪ʶ����ȡ�Ǩ�ơ����飬ȫ��ϸ�µ�˼�����ܵó���ȷ�Ľ��ۡ�

Ϊ��֤������Cl2��Fe3+��SO2��ijС������ͼ��ʾװ�ý���ʵ�飨�г������� A�м���װ�����ԣ��������Ѽ��飩��
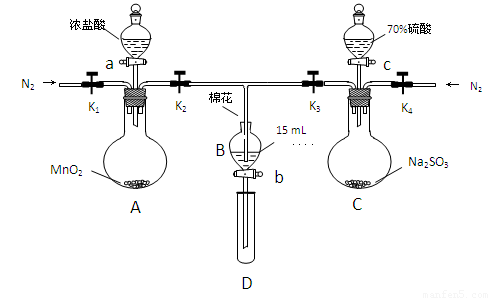
ʵ����̣�
��.���ɼ�K1~K4��ͨ��һ��ʱ��N2���ٽ�T�͵��ܲ���B�У�����ͨ��N2��Ȼ��ر�K1��K3��K4��
��.����a���μ�һ������Ũ���ᣬ��A���ȡ�
��.��B����Һ���ʱ��ֹͣ���ȣ��н����ɼ�K2��
��.����b��ʹԼ2mL����Һ����D�Թ��У��������е����ӡ�
��.���ɼ�K3������c������70%�����ᣬһ��ʱ���н����ɼ�K3��
��.�����Թ�D���ظ����̢�������B��Һ�е����ӡ�
��1��A�з�����Ӧ�Ļ�ѧ����ʽΪ ��
��2����70%��������ȡSO2����Ӧ���ʱ���98%������죬ԭ���� ��
��3���ס��ҡ�����λͬѧ�ֱ����������ʵ�飬���ǵļ����һ���ܹ�֤��������
Cl2��Fe3+��SO2���� ����ס����ҡ�����������
|
|
���̢� B��Һ�к��е����� |
���̢� B��Һ�к��е����� |
|
�� |
��Fe3+��Fe2+ |
��SO42- |
|
�� |
����Fe3+����Fe2+ |
��SO42- |
|
�� |
��Fe3+��Fe2+ |
��Fe2+ |
��4������ʵ����̢�ʱ��B����Һ��ɫ�ɻ�ɫ��Ϊ����ɫ��ֹͣͨ��������һ��ʱ�����Һ��ɫ��Ϊdz��ɫ��
�������ϣ�Fe2+��aq����SO32-��aq�� FeSO3��s����ī��ɫ��
FeSO3��s����ī��ɫ��
������裺FeCl3�� SO2�ķ�Ӧ�������м����FeSO3����Һ�ĺ���ɫ��FeSO3��ī��ɫ����FeCl3����ɫ���Ļ��ɫ��ijͬѧ�������ʵ�飬֤ʵ�ü��������

�� ��ҺE��F�ֱ�Ϊ �� ��
�� ���û�ѧƽ��ԭ�����Ͳ���3�к���ɫ��Һ��ɫ��Ϊdz��ɫ��ԭ��
��
Ϊ��֤������Cl2��Fe3����SO2��ijС������ͼ��ʾװ�ý���ʵ�飨�г������ͼ��м���װ�����ԣ��������Ѽ��飩��
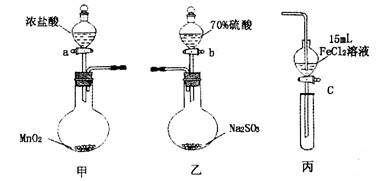
ʵ�鲽�裺
1.�ڼ�װ���У�����a�����ȣ���װ���г�������ɫ����ʱ�����װ�����ӡ�
2.����װ����FeC12��Һ���ʱ��ֹͣ���ȡ�
3.����c��ʹԼ2mL����Һ�����Թ��У�������Һ�е����ӡ�
4.����װ���У�����b���������ž������в���������ͨ��������װ�ñ�ƺ����Һ�У�һ��ʱ���ֹͣ��
5.���±����Թܣ�����c��ʹԼ2mL����Һ�����Թ��У�������Һ�е����ӡ�
�ش��������⣺
��1�����з�����Ӧ�Ļ�ѧ����ʽΪ______________________��
��2����70%��������ȡSO2����Ӧ���ʱ���98%������죬ԭ����____________��
��3��ʵ���У�֤��������Fe3+��SO2�����ӷ���ʽΪ_____________��
��4����I��II��III����ͬѧ�ֱ����������ʵ�飬ʵ�������£�
|
|
����3��Һ�к��е����� |
����5��Һ�к��е����� |
|
I |
����Fe3������Fe2�� |
��SO42�� |
|
�� |
��Fe3����Fe2�� |
��SO42�� |
|
�� |
��Fe3����Fe2�� |
��Fe2�� |
����ʵ����һ���ܹ�֤��Cl2��Fe3����SO2����______���I������II����III������
Ϊ��֤�����ԣ�Cl2��Fe3+��SO2��ijС������ͼ��ʾװ�ý���ʵ�飨�г�������A�еļ���װ�����ԣ��������Ѿ�������ϣ�
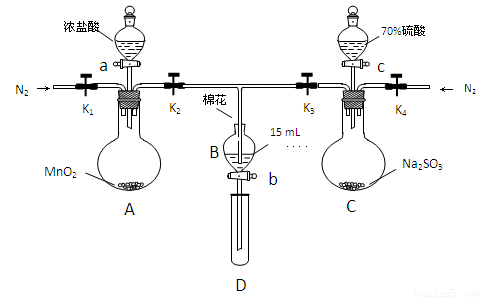
ʵ����̣�
���ɼ�K1-K4��ͨ��һ��ʱ��N2���ٽ�T�͵��ܲ���B�У�����ͨ��N2������ر�K1��K2��K3 .
����a���μ�һ������Ũ���ᣬ��A����.
��B�е���Һ���ʱ��ֹͣ���ȣ��н����ɼ�K2.
��������b��ʹԼ2ml����Һ����D�Թ��У��������е�����.
�������ɼ�K3������c������70%�����ᣬһ��ʱ���н����ɼ�K3.
��.�����Թ�D���ظ����̢�������B��Һ�е�����.
��1�����̢��Ŀ����___________________________
��2�����н������ҺΪ_______________________
��3��A�з�����Ӧ�Ļ�ѧ����ʽΪ_____________________________________________
��4����70%������֧ȡSO2����Ӧ���ʱ���98%������飬ԭ����___________________
��5�����̢��м���B��Һ���Ƿ���������IJ�����______________________________
��6���ס��ҡ�����λͬѧ�ֱ����������ʵ�飬���ǵļ����һ���ܹ�֤������Cl2��Fe3+��SO2����
|
|
���̢� B��Һ�к��е����� |
���̢� B��Һ�к��е����� |
|
�� |
��Fe3+��Fe2+ |
��SO42- |
|
�� |
����Fe3+����Fe2+ |
��SO42- |
|
�� |
��Fe3+��Fe2+ |
��Fe2+ |
��7������ʵ����̢�ʱ��B�е���Һ��ɫ�ɻ�ɫ��Ϊ����ɫ��ֹͣͨ��������һ��ʱ�����Һ��ɫ��Ϊdz��ɫ��
�������ϣ�Fe2+(aq)+SO32-(aq)
 FeSO3��s��
FeSO3��s��
������裺FeCl3��SO2�ķ�Ӧ�������м����FeSO3����Һ�ĺ���ɫ��FeSO3��ī��ɫ����FeCl3����ɫ���Ļ����ɫ��
ijͬѧ�������ʵ�飬��ʽ�ü���ij�����

��ҺE��F�ֱ���___________��_______________
���û�ѧƽ��ԭ�����Ͳ���3����ɫ��Һ��Ϊdz��ɫ��ԭ��___________________________
Ϊ��֤������Cl2 > Fe3+ > SO2��ijС������ͼ��ʾװ�ý���ʵ�飨�г�������A�м���װ�����ԣ��������Ѽ��飩��
ʵ����̣�
���ɼ�K1~K4��ͨ��һ��ʱ��N2���ٽ�T�͵��ܲ���B�У� ����ͨ��N2��Ȼ��ر�K1��K3��K4��
����a���μ�һ������Ũ���ᣬ����A��
��B����Һ���ʱ��ֹͣ���ȣ��н�K2��
��������b��ʹԼ2mL����Һ����D�Թ��У����������е����ӡ�
������K3�ͻ���c������70%�����ᣬһ��ʱ���н�K3��
���������Թ�D���ظ����̢���
��1�����̢��Ŀ���� ��
��2�����н������ҺΪ ��
��3��A�з�����Ӧ�Ļ�ѧ����ʽΪ ��
��4����˵��������Fe3+ > SO2�����ӷ���ʽ�� ��
��5�����̢�������B��Һ���Ƿ���SO42-�IJ����� ��
��6���ס��ҡ�����λͬѧ�ֱ����������ʵ�飬���ǵļ�������ܹ�֤��������Cl2 > Fe3+ > SO2���� ����ס����ҡ��������������ñ�Ҫ������˵������ ��
|
|
���̢� B��Һ�к��е����� |
���̢� B��Һ�к��е����� |
|
�� |
��Fe3+��Fe2+ |
�� Fe2+ |
|
�� |
����Fe3+����Fe2+ |
��SO42- |
|
�� |
��Fe3+��Fe2+ |
��SO42- |
��7������ʵ����̢�ʱ��B����Һ��ɫ�ɻ�ɫ��Ϊ����ɫ��ֹͣͨ��������һ��ʱ�����Һ��ɫ��Ϊdz��ɫ��
�������ϣ� Fe2+(aq)+
SO32- (aq)  FeSO3(s)��ī��ɫ��������裺FeCl3�� SO2�ķ�Ӧ�������м����FeSO3����Һ�ĺ���ɫ��FeSO3��ī��ɫ����FeCl3����ɫ���Ļ��ɫ��
FeSO3(s)��ī��ɫ��������裺FeCl3�� SO2�ķ�Ӧ�������м����FeSO3����Һ�ĺ���ɫ��FeSO3��ī��ɫ����FeCl3����ɫ���Ļ��ɫ��
ijͬѧ�������ʵ�飬֤ʵ�ü��������

�� ��ҺE��F�ֱ�Ϊ �� ��д��ѧʽ����
�����û�ѧƽ��ԭ�����Ͳ���3�к���ɫ��Һ��ɫ��Ϊdz��ɫ��ԭ�� ��