��Ŀ����
��ͼΪһ��������ͼ����С����ͼ���������ҷֱ�д��H2��CO2��Na2O��NaCl��FeCl3�������ʣ�ͼ���������������ʾ��ɹ���Ϊһ�࣬�ཻ����������A��B��C��DΪ����Ӧ�ķ������ݴ��š�

��ش��������⣺
(1)�뽫�������ݴ���������Ӧ��������
( )�������ʶ����ǵ����
( )�������ʶ����ƵĻ�����
( )�������ʶ���������
( )�������ʶ�����
(2)�ýྻ���ձ�ȡ��������ˮ���þƾ��Ƽ��������ڣ����ձ�����μ���
1 mol��L-1��ͼ��һ�ֻ������ˮ��Һ�����Ƶ�һ�ֺ��ɫ���塣
�ٷ�Ӧ�Ļ�ѧ����ʽΪ��________________________________________________��
������ýϼķ����жϽ����Ƿ��Ʊ��ɹ���
___________________________________________________________
����ý�������μ������ᣬ�����һϵ�б仯��
a���ȳ��ֺ��ɫ������ԭ����___________________________________________
b�������ɫ�����ܽ⣬�˷�Ӧ�����ӷ���ʽ��____________________________________

��ش��������⣺
(1)�뽫�������ݴ���������Ӧ��������
( )�������ʶ����ǵ����
( )�������ʶ����ƵĻ�����
( )�������ʶ���������
( )�������ʶ�����
(2)�ýྻ���ձ�ȡ��������ˮ���þƾ��Ƽ��������ڣ����ձ�����μ���
1 mol��L-1��ͼ��һ�ֻ������ˮ��Һ�����Ƶ�һ�ֺ��ɫ���塣
�ٷ�Ӧ�Ļ�ѧ����ʽΪ��________________________________________________��
������ýϼķ����жϽ����Ƿ��Ʊ��ɹ���
___________________________________________________________
����ý�������μ������ᣬ�����һϵ�б仯��
a���ȳ��ֺ��ɫ������ԭ����___________________________________________
b�������ɫ�����ܽ⣬�˷�Ӧ�����ӷ���ʽ��____________________________________
(1)A C B D
(2)��FeCl3��3H2O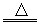 Fe(OH)3(����)��3HCl
Fe(OH)3(����)��3HCl
�����÷�ɢϵ�ܲ��������ЧӦ�����Ʊ��ɹ��������ɹ�
��a.�����HClʹFe(OH)3���巢���۳�����Fe(OH)3����
b��Fe(OH)3��3H�� =Fe3����3H2O
(2)��FeCl3��3H2O
 Fe(OH)3(����)��3HCl
Fe(OH)3(����)��3HCl�����÷�ɢϵ�ܲ��������ЧӦ�����Ʊ��ɹ��������ɹ�
��a.�����HClʹFe(OH)3���巢���۳�����Fe(OH)3����
b��Fe(OH)3��3H�� =Fe3����3H2O
(1)H2��CO2�����ǵ���ʣ�CO2��Na2O���������Na2O��NaCl�����ƵĻ����NaCl��FeCl3�����Ρ�(2)FeCl3��Һ�����ˮ�п��Ƶú��ɫ��Fe(OH)3���塣�����Ƴɵ��Ƿ��ǽ��壬���ö����ЧӦ

��ϰ��ϵ�д�
�����Ŀ