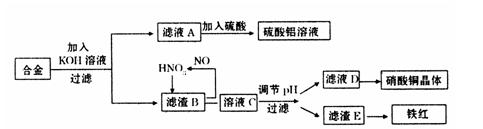
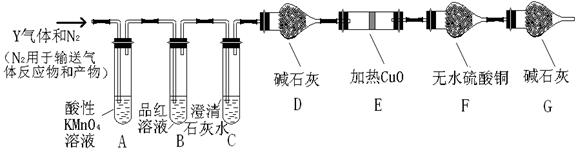
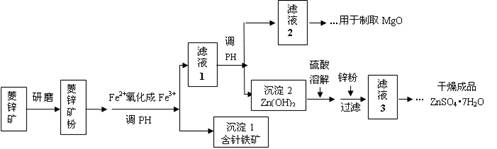
��Ŀ����
������ijͬѧ����250mL 0.2mol/L��NaOH��Һ��һЩ���裬����û�д������
| A����������ƽ���������зֱ�ŵ�������ֽ����ȡ2.0g NaOH���� |
| B����NaOH�������ձ����ܽ��Ѹ��С��ת����250mL����ƿ�� |
| C������ʱ��С�ļ�ˮ�����˿̶��ߣ���ʱѸ���ý�ͷ�ι�����һЩ |
| D����Һ������ϣ���������ת�������������Լ�ƿ�����ϱ�ǩ��ϴ������ƿ |
D
��

��ϰ��ϵ�д�
�����Ŀ