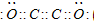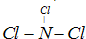��Ŀ����
����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������A��Dͬ���壬C��Eͬ���壬D��E��Fͬ���ڣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C�γɵ���������ȵķ��ӣ���A��C�γɵĻ����ﳣ����ΪҺ̬��A�ֱܷ���E��F�γɵ���������ȵ���̬���ӣ�
�Իش��������⣺
��1��E��F����̬�⻯����ȶ���Ϊ
��2��E��F���γ�E2F2�Ļ�����仯ѧʽΪ
��3��C��D�γɵ�һ�ֻ���������D��E�γɵĻ���������Һ�з���������ԭ��Ӧ�������ӷ���ʽΪ
��4��A��C��E����Ԫ���γɵ�һ�ֳ���������H����Ũ��Һ�ڼ��������¿���agͭ��Ӧ����ԭ��H�����ʵ���Ϊ
mol
mol��
�Իش��������⣺
��1��E��F����̬�⻯����ȶ���Ϊ
HCl
HCl
��H2S
H2S
���ѧʽ������2��E��F���γ�E2F2�Ļ�����仯ѧʽΪ
S2Cl2
S2Cl2
����3��C��D�γɵ�һ�ֻ���������D��E�γɵĻ���������Һ�з���������ԭ��Ӧ�������ӷ���ʽΪ
Na2O2+S2-+2H2O=S��+2Na++4OH-
Na2O2+S2-+2H2O=S��+2Na++4OH-
����4��A��C��E����Ԫ���γɵ�һ�ֳ���������H����Ũ��Һ�ڼ��������¿���agͭ��Ӧ����ԭ��H�����ʵ���Ϊ
| a |
| 64 |
| a |
| 64 |
������A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������A��C�γɵĻ����ﳣ����ΪҺ̬��������Ϊˮ����AΪH��CΪO��A��B������������֮����C��������������ȣ�B������������Ϊ5����BΪNԪ�أ�A��Dͬ���壬��DΪNa��C��Eͬ���壬��EΪS��D��E��Fͬ���ڣ�A�ֱܷ���E��F�γɵ���������ȵ�������ӣ���FΪCl��
��1���ǽ�����Խǿ����̬�⻯��Խ�ȶ���
��2��E2F2ΪS2Cl2���Թ��ۼ��γɵķ��ӣ�Ϊ���Ӿ��壻
��3��C��D�γɵ�һ�ֻ�����ΪNa2O2������ǿ�����ԣ�D��E�γɵĻ�����ΪNa2S�����߷�Ӧ����������������
��4��������HΪ���ᣬ�ɵ����غ������㣮
��1���ǽ�����Խǿ����̬�⻯��Խ�ȶ���
��2��E2F2ΪS2Cl2���Թ��ۼ��γɵķ��ӣ�Ϊ���Ӿ��壻
��3��C��D�γɵ�һ�ֻ�����ΪNa2O2������ǿ�����ԣ�D��E�γɵĻ�����ΪNa2S�����߷�Ӧ����������������
��4��������HΪ���ᣬ�ɵ����غ������㣮
����⣺A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������A��C�γɵĻ����ﳣ����ΪҺ̬��������Ϊˮ����AΪH��CΪO��A��B������������֮����C��������������ȣ�B������������Ϊ5����BΪNԪ�أ�A��Dͬ���壬��DΪNa��C��Eͬ���壬��EΪS��D��E��Fͬ���ڣ�A�ֱܷ���E��F�γɵ���������ȵ�������ӣ���FΪCl��
��1���ǽ�����Cl��S������̬�⻯����ȶ�ΪHCl��H2S���ʴ�Ϊ��HCl��H2S��
��2��EΪSԪ�ء�FΪClԪ�أ���E2F2ΪS2Cl2���ʴ�Ϊ��S2Cl2��
��3��C��D�γɵ�һ�ֻ�����ΪNa2O2������ǿ�����ԣ�D��E�γɵĻ�����ΪNa2S�����߷���������ԭ��Ӧ�������ӷ�ӦΪNa2O2+S2-+2H2O=S��+2Na++4OH-��
�ʴ�Ϊ��Na2O2+S2-+2H2O=S��+2Na++4OH-��
��4��������HΪ���ᣬCu��Ũ���ᷴӦ��Cuʧȥ���ӣ�S�õ����ӣ��豻��ԭ����������ʵ���Ϊx���ɵ����غ��֪��x����6-4��=
����2-0�������x=
mol��
�ʴ�Ϊ��
mol��
��1���ǽ�����Cl��S������̬�⻯����ȶ�ΪHCl��H2S���ʴ�Ϊ��HCl��H2S��
��2��EΪSԪ�ء�FΪClԪ�أ���E2F2ΪS2Cl2���ʴ�Ϊ��S2Cl2��
��3��C��D�γɵ�һ�ֻ�����ΪNa2O2������ǿ�����ԣ�D��E�γɵĻ�����ΪNa2S�����߷���������ԭ��Ӧ�������ӷ�ӦΪNa2O2+S2-+2H2O=S��+2Na++4OH-��
�ʴ�Ϊ��Na2O2+S2-+2H2O=S��+2Na++4OH-��
��4��������HΪ���ᣬCu��Ũ���ᷴӦ��Cuʧȥ���ӣ�S�õ����ӣ��豻��ԭ����������ʵ���Ϊx���ɵ����غ��֪��x����6-4��=
| ag |
| 64g/mol |
| a |
| 64 |
�ʴ�Ϊ��
| a |
| 64 |
���������⿼��λ�ýṹ���ʵĹ�ϵ��Ӧ�á�������ԭ��Ӧ�ȣ��Ѷ��еȣ�Ԫ�ص��ƶ��ǽ����Ĺؼ�����Ϥ����Ԫ�ػ���������ʣ�

��ϰ��ϵ�д�
�����Ŀ
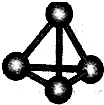 ����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��
����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��