��Ŀ����
ijѧ����NaHCO3��KHCO3��ɵĻ�������ʵ�飬����������ݣ�ÿ�μ������������ʵ���Ũ����ȣ������з�������ȷ���ǣ� ��
A����������ʵ���Ũ��Ϊ3.0mol/L
B�����ݱ��������ܼ���������NaHCO3����������
C����������9.2gʱ�������
D��15.7g�����ǡ����������ȫ��Ӧ
| ���� /mL | 50 | 50 | 50 |
| m������/g | 9.2 | 15.7 | 27.6 |
| V��CO2���������/L | 2.24 | 3.36L | 3.36L |
B�����ݱ��������ܼ���������NaHCO3����������
C����������9.2gʱ�������
D��15.7g�����ǡ����������ȫ��Ӧ
D
NaHCO3+HCl=NaCl+H2O+CO2����KHCO3+HCl=KCl+H2O+CO2����
A��ͨ��������������ݿ��Կ����������������л�����ǹ����ģ�����ȫ���μӷ�Ӧ��������ķ���ʽ���Կ�����������ʵ����Ͷ�����̼���ʵ�������ȵģ�
������̼���ʵ����ǣ�3.36/22.4=0.15mol��������������ʵ�������0.15mol��Ħ��Ũ�Ⱦ��ǣ�0.15/0.05=3mol/L����A��ȷ��
B����ǰ�����е����ݿ��Կ�����������̼�����������1/2������������Ҳ��Ҫ����1/2���ڶ�����ʵ�ʲμӷ�Ӧ�Ļ������13.8�ˣ�15.7�˵Ļ����û�з�Ӧ�꣬ʵ�������Ѿ������ˣ���̼��������x�ˣ���̼����ؾ��ǣ�13.8-x����
���ݷ���ʽ���Կ���̼�����ƺ�̼��������������ʵ�������1��1������̼���������ʵ����ǣ�x/84��̼��������ʵ����ǣ�(13.8-x)/100��x/84 +(13.8-x)/100=0.15����ã�x=6.3�ˣ�
��������Ϊ��6.3/13.8��100%=46%����B��ȷ
C����B�еļ�����Կ�����50ml�������13.8�˵Ļ���ﷴӦ������9.2��ʱ�����������C��ȷ��
D����ǰ�����е����ݿ��Կ�����������̼�����������1/2������������Ҳ��Ҫ����1/2���ڶ�����ʵ�ʲμӷ�Ӧ�Ļ������13.8�ˣ���D����
�ʴ�ΪD
A��ͨ��������������ݿ��Կ����������������л�����ǹ����ģ�����ȫ���μӷ�Ӧ��������ķ���ʽ���Կ�����������ʵ����Ͷ�����̼���ʵ�������ȵģ�
������̼���ʵ����ǣ�3.36/22.4=0.15mol��������������ʵ�������0.15mol��Ħ��Ũ�Ⱦ��ǣ�0.15/0.05=3mol/L����A��ȷ��
B����ǰ�����е����ݿ��Կ�����������̼�����������1/2������������Ҳ��Ҫ����1/2���ڶ�����ʵ�ʲμӷ�Ӧ�Ļ������13.8�ˣ�15.7�˵Ļ����û�з�Ӧ�꣬ʵ�������Ѿ������ˣ���̼��������x�ˣ���̼����ؾ��ǣ�13.8-x����
���ݷ���ʽ���Կ���̼�����ƺ�̼��������������ʵ�������1��1������̼���������ʵ����ǣ�x/84��̼��������ʵ����ǣ�(13.8-x)/100��x/84 +(13.8-x)/100=0.15����ã�x=6.3�ˣ�
��������Ϊ��6.3/13.8��100%=46%����B��ȷ
C����B�еļ�����Կ�����50ml�������13.8�˵Ļ���ﷴӦ������9.2��ʱ�����������C��ȷ��
D����ǰ�����е����ݿ��Կ�����������̼�����������1/2������������Ҳ��Ҫ����1/2���ڶ�����ʵ�ʲμӷ�Ӧ�Ļ������13.8�ˣ���D����
�ʴ�ΪD

��ϰ��ϵ�д�
 ���źþ���Ԫ����ĩ��ϵ�д�
���źþ���Ԫ����ĩ��ϵ�д� һ����ʦȨ����ҵ��ϵ�д�
һ����ʦȨ����ҵ��ϵ�д�
�����Ŀ
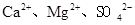 ���������ӣ�ʵ�����ᴿNaCl���������£�
���������ӣ�ʵ�����ᴿNaCl���������£�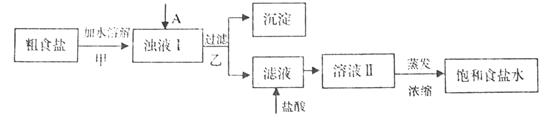
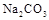 ��Һ����
��Һ����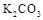 ��Һ
��Һ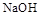 ��Һ
��Һ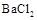 ��Һ
��Һ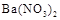 ��Һ
��Һ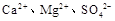 ���ӣ�ѡ��A���������Լ������μ�˳������Ϊ ��ֻ�ѧʽ����������Һ��
���ӣ�ѡ��A���������Լ������μ�˳������Ϊ ��ֻ�ѧʽ����������Һ��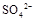 ������ȫ�IJ���Ϊ ��
������ȫ�IJ���Ϊ ��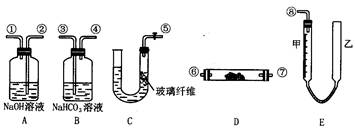
 �����Ʋ⣬ʵ�����б������������Ƶ���ȷ�����ǣ� ��
�����Ʋ⣬ʵ�����б������������Ƶ���ȷ�����ǣ� ��