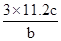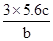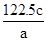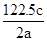��Ŀ����
��NAΪ�����ӵ���������ֵ������˵����ȷ����
A����֪N2(g)+3H2(g)  2NH3(g) ��H=��92.4kJ/mol���ڸ÷�Ӧ�У�����3NA������ת��ʱ�������Ȼ�ѧ����ʽ���ʱ��Ϊ����H=��46.2kJ/mol 2NH3(g) ��H=��92.4kJ/mol���ڸ÷�Ӧ�У�����3NA������ת��ʱ�������Ȼ�ѧ����ʽ���ʱ��Ϊ����H=��46.2kJ/mol |
| B����״���£�22.4L�������е������������ΪNA |
| C��1L0.1mol/L��CH3COOH��Һ�к���CH3COO��Ϊ0.1NA |
| D��1molFe��������ϡ�����ַ�Ӧ��ת�Ƶĵ�����ĿΪ2NA |
B
������������ݻ��ϼ�������������ת�Ƶ������ɵã�N2(g)+3H2(g)
 2NH3(g)��6 e�������ת�Ƶ�n(e��)=3mol����μӷ�Ӧ�ĵ��������ʵ������룬�����Ȼ�ѧ����ʽ�е�����ϵ����û�м��룬���Ը÷�Ӧ���ʱ�Ҳ������룬��H��Ϊ��92.4kJ/mol����A����n(����)=22.4L��22.4L/mol=1mol���������ɸ������������ɵĻ�������������ܵ����ʵ���Ϊ1mol����B��ȷ����Ȼn(CH3COOH)=0.1mol/L��1L=0.1mol�����Ǵ��������ᣬ��������ˮʱ������ȫ����Ϊ��������ӣ���n(CH3COO��)<0.1mol����C�����������ǿ�����ԣ��ܽ�����������������Ϊ���Σ���Fe+4HNO3==Fe(NO3)3+NO��+2H2O��3e��������ϵ��֮�ȵ������ʵ���֮�ȿ�֪��1molFe��ȫ��Ӧת��3mol���ӣ���D����
2NH3(g)��6 e�������ת�Ƶ�n(e��)=3mol����μӷ�Ӧ�ĵ��������ʵ������룬�����Ȼ�ѧ����ʽ�е�����ϵ����û�м��룬���Ը÷�Ӧ���ʱ�Ҳ������룬��H��Ϊ��92.4kJ/mol����A����n(����)=22.4L��22.4L/mol=1mol���������ɸ������������ɵĻ�������������ܵ����ʵ���Ϊ1mol����B��ȷ����Ȼn(CH3COOH)=0.1mol/L��1L=0.1mol�����Ǵ��������ᣬ��������ˮʱ������ȫ����Ϊ��������ӣ���n(CH3COO��)<0.1mol����C�����������ǿ�����ԣ��ܽ�����������������Ϊ���Σ���Fe+4HNO3==Fe(NO3)3+NO��+2H2O��3e��������ϵ��֮�ȵ������ʵ���֮�ȿ�֪��1molFe��ȫ��Ӧת��3mol���ӣ���D����
��ϰ��ϵ�д�
 ������ȫ�̼����ĩ���100��ϵ�д�
������ȫ�̼����ĩ���100��ϵ�д�
�����Ŀ