��Ŀ����
 A��B��C��D��Ϊ��ѧ��ѧ�������ʣ�����֮��ķ�Ӧ��ϵ��ͼ��ʾ��
A��B��C��D��Ϊ��ѧ��ѧ�������ʣ�����֮��ķ�Ӧ��ϵ��ͼ��ʾ��
��1����A��̼���ƣ�A��B��ˮ��Һ��Ӧʱֻ��������C��CaCO3��H2O����B��ֻ������Ԫ�أ��Ļ�ѧʽΪ______�����̬ʱ�ľ���Ϊ______���壻C�����к��еĻ�ѧ��������______��
��2����A�Ƕ�������ԭ�Ӱ뾶����Ԫ�صĵ��ʣ�D�ȿ�����NaOH��Һ�ֿ��������ᣮ��д����
��D������������ӷ���ʽ______��
�ڹ�ҵ��ұ��A�Ļ�ѧ����ʽ______��
��ʵ�����ö��Ե缫���100mL 0.1mol/L��A���Ȼ����ˮ��Һ�����з�̪����һ��ʱ����������������õ�112mL���壨��״��������������Һ��pHΪ______�����Է�Ӧǰ����Һ������仯����ij�缫������Һ��ɫ��죬ԭ����______����õ缫�����ֱ����Դ��______�������ӣ����������������
��3����A�ǿ�����ǿ�B�����Σ�D������ϡ���ᣬ��Ӧ�Ļ�ѧ����ʽ��______��
��4����A������Ϊ����ɫ���壬B�Ǻ������Σ�D���������ᣬ��D�Ļ�ѧʽ��______��
�⣺��1��̼������ˮ����ҷ�Ӧ�����������ƺ���Ȳ���壬���ɵ�������������B������Ӧ��������̼��Ƴ�����Bֻ������Ԫ�أ�ֻ��ΪCO2����̬CO2Ϊ���Ӿ��壬CΪCH��CH�����м��Լ��ͷǼ��Լ���
�ʴ�Ϊ��CO2�����ӣ����Լ��ͷǼ��Լ���
��2����A�Ƕ�������ԭ�Ӱ뾶����Ԫ�صĵ��ʣ�ӦΪNa����ҵ�õ��NaCl�ķ���ұ����D�ȿ�����NaOH��Һ�ֿ��������ᣬӦΪAl��OH��3��
��Al��OH��3�����ᷴӦ����Al3+����Ӧ�����ӷ���ʽΪAl��OH��3+3H+=Al3++3H2O��
�ʴ�Ϊ��Al��OH��3+3H+=Al3++3H2O��
�ڹ�ҵ�õ�����ڵ�NaCl�ķ���ұ��Na������ʽΪ2NaCl 2Na+Cl2�����ʴ�Ϊ��2NaCl
2Na+Cl2�����ʴ�Ϊ��2NaCl 2Na+Cl2����
2Na+Cl2����
�۵��NaCl��Һ������ӦΪ��������2Cl--2e-=Cl2����������2H++2e-=H2�����������õ�112mL���壬��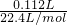 =0.005mol��Cl-�պ÷�Ӧ�꣬����2��0.005mol=0.01molOH-����������Һ��pHΪ13���������Դ�ĸ��������ӣ��������������õ���������������ˮ�ĵ���ƽ�ⱻ�ƻ���ʹ��������������������Ũ�ȴ���������Ũ�ȣ��缫������Һ��ɫ��죬
=0.005mol��Cl-�պ÷�Ӧ�꣬����2��0.005mol=0.01molOH-����������Һ��pHΪ13���������Դ�ĸ��������ӣ��������������õ���������������ˮ�ĵ���ƽ�ⱻ�ƻ���ʹ��������������������Ũ�ȴ���������Ũ�ȣ��缫������Һ��ɫ��죬
�ʴ�Ϊ��13���������������õ���������������ˮ�ĵ���ƽ�ⱻ�ƻ���ʹ��������������������Ũ�ȴ���������Ũ�ȣ�����
��3��A����ǿ�Ӧ�����壬��AΪ��Σ���D������ϡ�������֪DΪ���ᱵ��������֪BΪ����泥���Ӧ�ķ���ʽΪBa��OH��2+��NH4��2SO4=BaSO4��+2NH3��+2H2O��
�ʴ�Ϊ��Ba��OH��2+��NH4��2SO4=BaSO4��+2NH3��+2H2O��
��4��A������Ϊ����ɫ���壬ӦΪNO2����ˮ��Ӧ����NO�����ᣬB�Ǻ������Σ������ᷴӦ���ɺ����ᣬD���������ᣬӦΪH2SiO3��
�ʴ�Ϊ��H2SiO3��
��������1��̼������ˮ����ҷ�Ӧ�����������ƺ���Ȳ���壬���ɵ�������������B������Ӧ��������̼��Ƴ�����Bֻ������Ԫ�أ�ֻ��ΪCO2��
��2����A�Ƕ�������ԭ�Ӱ뾶����Ԫ�صĵ��ʣ�ӦΪNa����ҵ�õ��NaCl�ķ���ұ����D�ȿ�����NaOH��Һ�ֿ��������ᣬӦΪAl��OH��3��
��3��A����ǿ�Ӧ�����壬��AΪ��Σ���D������ϡ�������֪DΪ���ᱵ��������֪BΪ����泥�
��4��A������Ϊ����ɫ���壬ӦΪNO2����ˮ��Ӧ����NO�����ᣬB�Ǻ������Σ������ᷴӦ���ɺ����ᣬD���������ᣬӦΪH2SiO3��
���������⿼��������ƶϣ���Ŀ�Ѷ��еȣ������״���Ϊ��2����ע�����ԭ���ԭ��������ע��������ʵ����ʽ�Ϸ�Ӧ��������ƶϣ�ѧϰ��ע����ػ���֪ʶ�Ļ��ۣ�
�ʴ�Ϊ��CO2�����ӣ����Լ��ͷǼ��Լ���
��2����A�Ƕ�������ԭ�Ӱ뾶����Ԫ�صĵ��ʣ�ӦΪNa����ҵ�õ��NaCl�ķ���ұ����D�ȿ�����NaOH��Һ�ֿ��������ᣬӦΪAl��OH��3��
��Al��OH��3�����ᷴӦ����Al3+����Ӧ�����ӷ���ʽΪAl��OH��3+3H+=Al3++3H2O��
�ʴ�Ϊ��Al��OH��3+3H+=Al3++3H2O��
�ڹ�ҵ�õ�����ڵ�NaCl�ķ���ұ��Na������ʽΪ2NaCl
 2Na+Cl2�����ʴ�Ϊ��2NaCl
2Na+Cl2�����ʴ�Ϊ��2NaCl 2Na+Cl2����
2Na+Cl2�����۵��NaCl��Һ������ӦΪ��������2Cl--2e-=Cl2����������2H++2e-=H2�����������õ�112mL���壬��
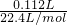 =0.005mol��Cl-�պ÷�Ӧ�꣬����2��0.005mol=0.01molOH-����������Һ��pHΪ13���������Դ�ĸ��������ӣ��������������õ���������������ˮ�ĵ���ƽ�ⱻ�ƻ���ʹ��������������������Ũ�ȴ���������Ũ�ȣ��缫������Һ��ɫ��죬
=0.005mol��Cl-�պ÷�Ӧ�꣬����2��0.005mol=0.01molOH-����������Һ��pHΪ13���������Դ�ĸ��������ӣ��������������õ���������������ˮ�ĵ���ƽ�ⱻ�ƻ���ʹ��������������������Ũ�ȴ���������Ũ�ȣ��缫������Һ��ɫ��죬�ʴ�Ϊ��13���������������õ���������������ˮ�ĵ���ƽ�ⱻ�ƻ���ʹ��������������������Ũ�ȴ���������Ũ�ȣ�����
��3��A����ǿ�Ӧ�����壬��AΪ��Σ���D������ϡ�������֪DΪ���ᱵ��������֪BΪ����泥���Ӧ�ķ���ʽΪBa��OH��2+��NH4��2SO4=BaSO4��+2NH3��+2H2O��
�ʴ�Ϊ��Ba��OH��2+��NH4��2SO4=BaSO4��+2NH3��+2H2O��
��4��A������Ϊ����ɫ���壬ӦΪNO2����ˮ��Ӧ����NO�����ᣬB�Ǻ������Σ������ᷴӦ���ɺ����ᣬD���������ᣬӦΪH2SiO3��
�ʴ�Ϊ��H2SiO3��
��������1��̼������ˮ����ҷ�Ӧ�����������ƺ���Ȳ���壬���ɵ�������������B������Ӧ��������̼��Ƴ�����Bֻ������Ԫ�أ�ֻ��ΪCO2��
��2����A�Ƕ�������ԭ�Ӱ뾶����Ԫ�صĵ��ʣ�ӦΪNa����ҵ�õ��NaCl�ķ���ұ����D�ȿ�����NaOH��Һ�ֿ��������ᣬӦΪAl��OH��3��
��3��A����ǿ�Ӧ�����壬��AΪ��Σ���D������ϡ�������֪DΪ���ᱵ��������֪BΪ����泥�
��4��A������Ϊ����ɫ���壬ӦΪNO2����ˮ��Ӧ����NO�����ᣬB�Ǻ������Σ������ᷴӦ���ɺ����ᣬD���������ᣬӦΪH2SiO3��
���������⿼��������ƶϣ���Ŀ�Ѷ��еȣ������״���Ϊ��2����ע�����ԭ���ԭ��������ע��������ʵ����ʽ�Ϸ�Ӧ��������ƶϣ�ѧϰ��ע����ػ���֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
 53���ò�ϵ�д�
53���ò�ϵ�д�
�����Ŀ
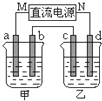 ��ͼ��ʾ��װ���У��ס������ձ��зֱ�ʢ����������CuSO4��Һ��100g 10.00%��K2SO4��Һ��a��b��c��d��Ϊʯī�缫����ͨ��Դһ��ʱ��������K2SO4��ҺŨ��Ϊ10.47%������a�缫���������ӣ�����˵����ȷ���ǣ�������
��ͼ��ʾ��װ���У��ס������ձ��зֱ�ʢ����������CuSO4��Һ��100g 10.00%��K2SO4��Һ��a��b��c��d��Ϊʯī�缫����ͨ��Դһ��ʱ��������K2SO4��ҺŨ��Ϊ10.47%������a�缫���������ӣ�����˵����ȷ���ǣ�������| A���ס�����Һ��pH����С | B���缫b��������������ԼΪ2.8L����״���£� | C���缫d�Ϸ����ķ�ӦΪ��2H2O+2e-?H2��+2OH- | D����ʹ���е���Һ�ָ���ԭ����Ũ�ȣ��ɼ���24.5g��Cu��OH��2 |
 A��B��C��D��Ϊ�������ʣ��֮��Ĺ�ϵ��ͼ��ʾ����-����ʾ�������ʼ��ܷ�����Ӧ����������ʾ���ʼ��ת����ϵ�����ַ�Ӧ����������Լ���Ӧ��������ȥ����
A��B��C��D��Ϊ�������ʣ��֮��Ĺ�ϵ��ͼ��ʾ����-����ʾ�������ʼ��ܷ�����Ӧ����������ʾ���ʼ��ת����ϵ�����ַ�Ӧ����������Լ���Ӧ��������ȥ����
 A��B��C��D��Ϊ��ѧ��ѧ�г��������ʣ�����֮���ת����ϵ����ͼ�����ֲ�������ȥ�����Իش�
A��B��C��D��Ϊ��ѧ��ѧ�г��������ʣ�����֮���ת����ϵ����ͼ�����ֲ�������ȥ�����Իش�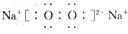
 A��B��C��D��Ϊ������Ԫ�أ�A��B��ͬ�������ڵ�����Ԫ�أ�A��C��ͬ�������ڵ�����Ԫ�أ�A��B��C����Ԫ�ص�ԭ������֮��Ϊ31��DԪ����A��B��C����Ԫ�ؼȲ���ͬ���ڣ�Ҳ��ͬ���壮��ش�
A��B��C��D��Ϊ������Ԫ�أ�A��B��ͬ�������ڵ�����Ԫ�أ�A��C��ͬ�������ڵ�����Ԫ�أ�A��B��C����Ԫ�ص�ԭ������֮��Ϊ31��DԪ����A��B��C����Ԫ�ؼȲ���ͬ���ڣ�Ҳ��ͬ���壮��ش�
 NH3?H2O+H+
NH3?H2O+H+ 2NH3
2NH3
 ���ͱ����γ����ֻ�����X��Y��X��ˮ��Ӧ������һ�־��л�ԭ�ԵĶ�Ԫ��M��1mol�����������ɻ�������Z�����õ�Z����ˮ��Ӧ�IJ���W����12mol KOH������ȫ�кͣ�������������ȼ�����ɻ�����N��N��ˮ��Ӧ����W��DԪ�ص���̬�⻯��Իش��������⣺
���ͱ����γ����ֻ�����X��Y��X��ˮ��Ӧ������һ�־��л�ԭ�ԵĶ�Ԫ��M��1mol�����������ɻ�������Z�����õ�Z����ˮ��Ӧ�IJ���W����12mol KOH������ȫ�кͣ�������������ȼ�����ɻ�����N��N��ˮ��Ӧ����W��DԪ�ص���̬�⻯��Իش��������⣺