��Ŀ����
3��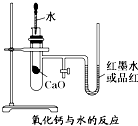 ��ͼ��ijͬѧ��Ƶķ��ȷ�Ӧ�Ĺ۲�װ�ã�
��ͼ��ijͬѧ��Ƶķ��ȷ�Ӧ�Ĺ۲�װ�ã���ʵ������Ǣٰ�ͼ��ʾ��ʵ��װ�����Ӻã�����U�ι��ڼ���������īˮ����T�ι������У�ʹU�ι������ߵ�Һ�洦��ͬһˮƽ�棬�ټн������У������м���Թ���ʢ1g�����ƣ�������2mL���ҵ�����ˮ���ɹ۲죮
�Իش�
��1��Ԥ��ʵ�����ܹ۲쵽��������U�β�������ĺ�īˮ����Ʒ�죩���ؿ��ڶ�������
��2����ʵ���Т٢ڲ���֮�������е�һ��ʵ������Ǽ��װ�������ԣ�
��3��ʵ���з����Ļ�ѧ��Ӧ����ʽCaO+H2O�TCa��OH��2��
��4��˵��CaO��H2O��������Ca��OH��2����֮��Ĺ�ϵ1molCaO��1molH2O�������ʹ���1molCa��OH��2��������
���� ��1�������ƺ�ˮ֮��ķ�Ӧ�Ƿ��ȵģ���������������������ʣ�
��2��������ѹԭ���µ�ʵ������һ��Ҫ��֤װ�ò�©����
��3��CaO��ˮ��Ӧ�Ļ�ѧ��Ӧ����Ca��OH��2��
��4�����ݷ�Ӧ����������������������֮��Ĵ�С��ϵ������Ӧ�������������
��� �⣺��1�������ƺ�ˮ֮��ķ�Ӧ�Ƿ��ȵģ������Թ����¶����ߣ���������������������ʣ������ڲ�ѹǿ����U�β�������ĺ�īˮ����Ʒ�죩���ؿ��ڶ�������
�ʴ�Ϊ��U�β�������ĺ�īˮ����Ʒ�죩���ؿ��ڶ�������
��2����ʵ����������ѹԭ���µ�ʵ��������֣�����ʵ��֮ǰһ��Ҫ���װ�������ԣ�
�ʴ�Ϊ�����װ�������ԣ�
��3��CaO��ˮ��Ӧ�����������ƣ���Ӧ�Ļ�ѧ��Ӧ����ʽΪ��CaO+H2O�TCa��OH��2��
�ʴ�Ϊ��CaO+H2O�TCa��OH��2��
��5��CaO+H2O�TCa��OH��2������ʵ�������֪�������ƺ�ˮ֮��ķ�Ӧ�Ƿ��ȵģ�1molCaO��1molH2O�������ʹ���1molCa��OH��2��������
�ʴ�Ϊ��1molCaO��1molH2O�������ʹ���1molCa��OH��2��������
���� ����ͨ��ʵ����ʽ̽����ѧ��Ӧ�е������仯����Ŀ�Ѷ��еȣ�ע����ȷ��ѧ��Ӧ�������仯�Ĺ�ϵ��Ҫ��ѧ��ѧ�����ѧ֪ʶ��Ǩ�ƺ�Ӧ�ã�����������ѧ���ķ�����������������ѧʵ��������

| A�� | -241.8 kJ•mol-1 | B�� | -20.6kJ•mol-1 | C�� | +29.7kJ•mol-1 | D�� | +99.7 kJ•mol-1 |
| A�� | FeBr2��Һ�е������������2Fe2++2Br-+2Cl2=2Fe3++Br2+4Cl- | |
| B�� | ���������£�FeSO4��Һ��H2O2��Ӧ��Fe2++2H++H2O2=Fe3++2H2O | |
| C�� | SO2ͨ��Ca��ClO��2��Һ�У�Ca��ClO��2+SO2+H2O=CaSO3��+2HClO | |
| D�� | NaAlO2��Һ��NaHCO3��Һ��ϣ�AlO2-+HCO3-+H2O=Al��OH��3��+CO32- |
| A�� | ���Ȼ�̼�ĵ���ʽ��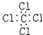 | |
| B�� | þ��ԭ�ӽṹʾ��ͼΪ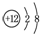 | |
| C�� | �Ȼ�����ӵ��γɹ��̿��õ���ʽ��ʾΪ��H��+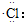 ��H+[ ��H+[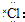 ]- ]- | |
| D�� | ��ˮ�Ļ�ѧʽΪ${\;}_{1}^{2}$H2O����D2O�� |
| A�� | 7�� | B�� | 9�� | C�� | 12�� | D�� | 14�� |
| A�� | 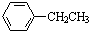 | B�� | HC��C-CH2CH3 | C�� | 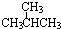 | D�� | 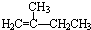 |
| A�� | C2H4+3O2$\stackrel{��ȼ}{��}$2CO2+2H2O | |
| B�� | H2+Cl2$\stackrel{����}{��}$2HCl | |
| C�� | CH2=CH2+Br2�� | |
| D�� | CH3-CH3+2Cl2$\stackrel{����}{��}$CH2Cl-CH2Cl+2HCl |
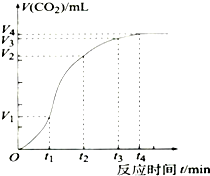 Ϊ���о�̼�����ϡ���ᷴӦ�ķ�Ӧ���ʣ�һλͬѧͨ��ʵ��ⶨ��Ӧ�����ɵ�CO2��������淴Ӧʱ��仯��������������ߣ�ͼ��������������������⣮
Ϊ���о�̼�����ϡ���ᷴӦ�ķ�Ӧ���ʣ�һλͬѧͨ��ʵ��ⶨ��Ӧ�����ɵ�CO2��������淴Ӧʱ��仯��������������ߣ�ͼ��������������������⣮