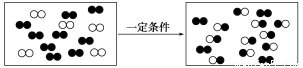
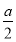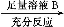��Ŀ����
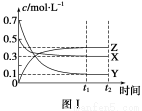
���ú�̼������ϳ�ȼ���ǽ����ԴΣ������Ҫ��������֪CO(g)��2H2(g)  CH3OH(g)��Ӧ�����е������仯�����ͼ��ʾ�����������������ֱ��ʾ��ʹ�ô�����ʹ�ô�������������������ж���ȷ���ǣ� ��
CH3OH(g)��Ӧ�����е������仯�����ͼ��ʾ�����������������ֱ��ʾ��ʹ�ô�����ʹ�ô�������������������ж���ȷ���ǣ� ��
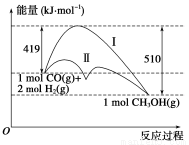
A���÷�Ӧ����H����91 kJ��mol��1
B������������÷�Ӧ����H��С
C����Ӧ��������������������������
D������÷�Ӧ����Һ̬CH3OH������H����
C
������������ͼʾ���÷�Ӧ��Ӧ�����������������������������Ƿ��ȷ�Ӧ����ѡ��A����C��ȷ���������ֻ�ܽ��ͷ�Ӧ����Ļ�ܣ����Է�Ӧ����Ӱ�죬ѡ��B��������Һ̬CH3OHʱ�ͷų����������࣬��H��С��ѡ��D����

������Դ�Ŀ��������þ��й�����ǰ������ˮ��pHһ����7.5��8.6֮�䡣ij�غ�ˮ����Ҫ���ӵĺ������±���
�ɷ� | Na�� | K�� | Ca2�� | Mg2�� | Cl�� | SO42- | HCO3- |
����/mg��L��1 | 9 360 | 83 | 200 | 1 100 | 16 000 | 1 200 | 118 |
��1�����������ǽ��귢չ������һ�ֽϺõĺ�ˮ������������ԭ������ͼ��ʾ��������(��)���ӽ���Ĥֻ������(��)����ͨ����
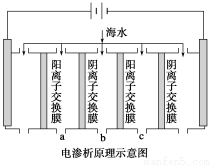
�������ĵ缫��ӦʽΪ__________________��
�����һ��ʱ�䣬�����������ˮ������ɷ�ΪCaCO3��Mg(OH)2��д������CaCO3�����ӷ���ʽ______________________________________��
����ˮ�ij���Ϊa��b��c�е�________���ڡ�
��2����ˮ���Ԫ�ش����dz��ḻ���Ӻ�ˮ����ȡ﮵��о�����DZ����������컯ѧ��Դ����Ҫԭ�ϣ���LiFePO4���ij�缫�Ĺ���ԭ������ͼ��ʾ��
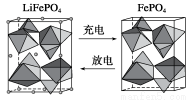
�õ�ص����Ϊ����Li���Ĺ�����ϡ��ŵ�ʱ�õ缫�ǵ�ص�________��(����������������)���缫��ӦʽΪ______________________��