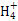��Ŀ����
ij�о�С������ͼװ�ý���SO2��FeCl3��Һ��Ӧ�����ʵ�飨�г�װ������ȥ����
��1���������Ȼ�����Һʱ�����Ȱ��Ȼ��������ܽ���________�У��ټ�ˮϡ�ͣ�����������Ŀ����________�������в���Ҫ��������________��������ȷѡ��ǰ����ĸ����
a��ҩ�ס� b���ձ��� c��ʯ������ d���������� e������
��2��ͨ������SO2ʱ��C�й۲쵽������Ϊ______________________________��
��3��������������С��ͬѧ��ΪSO2��FeCl3��Һ������������ԭ��Ӧ��
��д��SO2��FeCl3��Һ��Ӧ�����ӷ���ʽ��___________________________��
�������ʵ�鷽��������Fe2�����ɣ�__________________________________��
�۸�С��ͬѧ��C�Թܷ�Ӧ�����Һ�м��������ữ��BaCl2��Һ�������ְ�ɫ����������֤����Ӧ������SO42-��������________���������������������������_______________________________________________________________��
��4��Dװ���е���©����������______________________________________��
��5��Ϊ����֤SO2���л�ԭ�ԣ�ʵ���п��Դ���FeCl3���Լ���________��������ȷѡ��ǰ����ĸ����
a��ŨH2SO4 b������KMnO4��Һ
c����ˮ d��NaCl��Һ

��1���������Ȼ�����Һʱ�����Ȱ��Ȼ��������ܽ���________�У��ټ�ˮϡ�ͣ�����������Ŀ����________�������в���Ҫ��������________��������ȷѡ��ǰ����ĸ����
a��ҩ�ס� b���ձ��� c��ʯ������ d���������� e������
��2��ͨ������SO2ʱ��C�й۲쵽������Ϊ______________________________��
��3��������������С��ͬѧ��ΪSO2��FeCl3��Һ������������ԭ��Ӧ��
��д��SO2��FeCl3��Һ��Ӧ�����ӷ���ʽ��___________________________��
�������ʵ�鷽��������Fe2�����ɣ�__________________________________��
�۸�С��ͬѧ��C�Թܷ�Ӧ�����Һ�м��������ữ��BaCl2��Һ�������ְ�ɫ����������֤����Ӧ������SO42-��������________���������������������������_______________________________________________________________��
��4��Dװ���е���©����������______________________________________��
��5��Ϊ����֤SO2���л�ԭ�ԣ�ʵ���п��Դ���FeCl3���Լ���________��������ȷѡ��ǰ����ĸ����
a��ŨH2SO4 b������KMnO4��Һ
c����ˮ d��NaCl��Һ
��1��Ũ���ᡡ�����Ȼ���ˮ�⡡ce
��2����Һ���ػ�ɫ��Ϊdz��ɫ
��3����2Fe3����SO2��2H2O=SO42-��4H����2Fe2��
��ȡC�з�Ӧ�����Һ�������е���KSCN��Һ������죬�ټ������Ƶ���ˮ����H2O2��Һ�ȣ�����Һ���
�۲�������������Խ��ܽ��SO2����ΪH2SO4������ʵ��
��4��ʹ���������գ���ֹ��Һ����
��5��bc
��2����Һ���ػ�ɫ��Ϊdz��ɫ
��3����2Fe3����SO2��2H2O=SO42-��4H����2Fe2��
��ȡC�з�Ӧ�����Һ�������е���KSCN��Һ������죬�ټ������Ƶ���ˮ����H2O2��Һ�ȣ�����Һ���
�۲�������������Խ��ܽ��SO2����ΪH2SO4������ʵ��
��4��ʹ���������գ���ֹ��Һ����
��5��bc
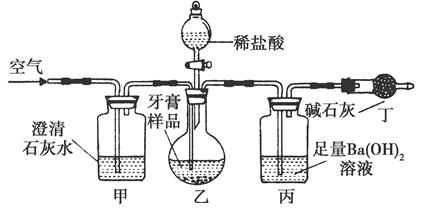
��1���Ȼ�����ˮ��Һ������ˮ�⣬��������Ȼ�����Һʱ��һ���Ȱ��Ȼ�����������Ũ�����У��ټ�ˮϡ�ͣ���������ˮ�⡣������Һ���ձ��н��У�����Ҫʯ��������������2��SO2��FeCl3��Һ����������ԭ��Ӧ��Fe3������ԭΪFe2������Һ���ػ�ɫ��Ϊdz��ɫ����3���ڼ���Fe2��ʱҪ�ų�Fe3���ĸ��ţ����ȼ�KSCN��Һ����ȷ����Һ�в�����Fe3��������£�������ˮ��������������Fe2��������Һ��졣��4�����õ�©����ˮ�нϴ�ĽӴ��������ʹ���������ղ���ֹ��Һ��������5����֤SO2���л�ԭ�ԣ�Ҫ���þ��������Ե��Լ�������SO2����������ԭ��Ӧ������������

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ



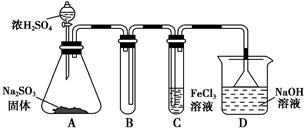
 Fe(SCN)3+3KCl��Һƽ����ϵ�м�����������KCl
Fe(SCN)3+3KCl��Һƽ����ϵ�м�����������KCl