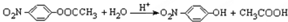��Ŀ����
��֪�����������������·�Ӧ����2BrO3����I2===2IO3����Br2����2IO3����10Br����12H��===I2��5Br2��6H2O��
��ش��������⣺
(1)��ͬѧ���ݷ�Ӧ�ٵó������ԣ�I2>Br2�Ľ��ۣ�����Ϊ�Ƿ���ȷ��______________________________
(����Ϊ��ȷ�����ȷ��������Ϊ����ȷ�������ȷ����˵������)��
(2)����6 mol KI��������Һ����μ���KBrO3��Һ�����������к������ʵ����ʵ�����������KBrO3�����ʵ�������ͼ��ʾ��ϵ��
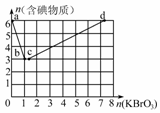
��a��b�εķ�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ________��b��ʱ��KIȫ�����뷴Ӧ����b�㴦��Ӧ�ĺ������ʵĻ�ѧʽΪ________��
��b��c�εķ�Ӧ����һ��Ԫ�صļ�̬�����˱仯���˽η�Ӧ�����ӷ���ʽΪ________��
��n(KBrO3)��4ʱ����Ӧ�������ʵĻ�ѧʽΪ________��
�ܺ�a mol KI��������Һ��������KBrO3���ʵ��������ֵΪ________��
(1)����ȷ����Ϊ�ڷ�Ӧ����I2����ԭ����Br2Ϊ��ԭ���ֻ�ܵó���ԭ��I2>Br2�Ľ���
(2)��1��6��I2
��5Br����BrO3����6H��===3Br2��3H2O
��I2��KIO3����1.2a mol

 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д� �������ϵ�д�
�������ϵ�д�
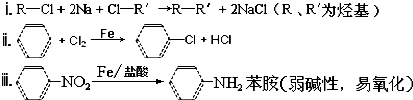
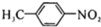
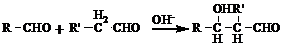 ��R��R'������������ԭ�ӣ�
��R��R'������������ԭ�ӣ�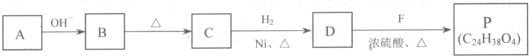
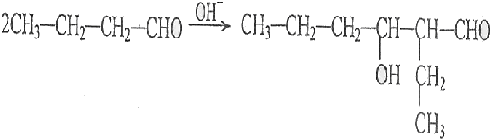
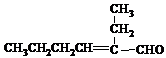
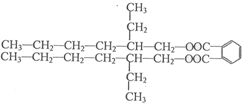
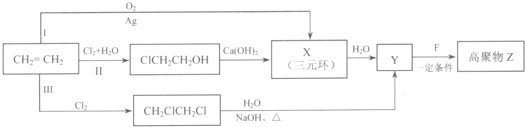
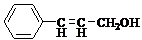 ���ĺϳ�·�ߣ�
���ĺϳ�·�ߣ�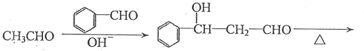
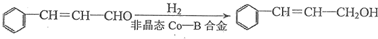
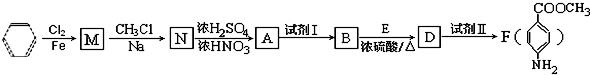
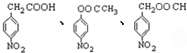 ����2��
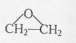
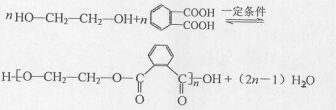
����2��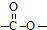 �ṹ
�ṹ