��Ŀ����
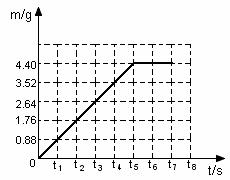
С��ͬѧ��13.9g�����ʵĵĴ�����Ʒ��̼�������Ȼ��ƵĻ�����90.5gϡ�������ϣ���ַ�Ӧ����÷�Ӧ���������������m���뷴Ӧʱ�䣨t�����������±���ʾ��| ��Ӧʱ��t/s | t0 | t1 | t2 | t3 | t4 | t5 | t6 |
| ��������m/g | 0 | 0.88 | 1.76 | 2.64 | 3.52 | 4.4 | 4.4 |
��1��̼������ȫ��Ӧ������CO2������Ϊ g
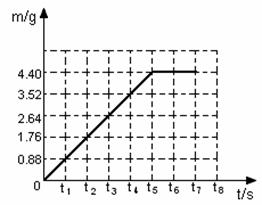
��2���������������ͼ�У�������Ӧʱ���������������m����ʱ�䣨t���仯�����ߡ�
��3������ȫ��Ӧ��������Һ�����ʵ�������������Na2CO3��2HCl��2NaCl��CO2����H2O����
��1��4.4
��2��
��3���⣺����Ʒ��̼���Ƶ�����Ϊx����Ӧ����NaCl������Ϊy��
Na2CO3��2HCl��2NaCl��CO2����H2O
106 117 4.4
x y 4.4
��x��10.6��g�� y��11.7��g��
����Ʒ���Ȼ��Ƶ�����Ϊ13.9��10.6��3.3��g��
��Ӧ����Һ�����ʵ�������11.7��3.3��15��g��
��Ӧ����Һ��������13.9��90.5��4.4��100��g��
����ȫ��Ӧ��������Һ�����ʵ���������Ϊ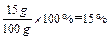
����ȫ��Ӧ��������Һ�����ʵ���������Ϊ15%����:
��
��2��

��3���⣺����Ʒ��̼���Ƶ�����Ϊx����Ӧ����NaCl������Ϊy��
Na2CO3��2HCl��2NaCl��CO2����H2O
106 117 4.4
x y 4.4
��x��10.6��g�� y��11.7��g��
����Ʒ���Ȼ��Ƶ�����Ϊ13.9��10.6��3.3��g��
��Ӧ����Һ�����ʵ�������11.7��3.3��15��g��
��Ӧ����Һ��������13.9��90.5��4.4��100��g��
����ȫ��Ӧ��������Һ�����ʵ���������Ϊ
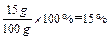
����ȫ��Ӧ��������Һ�����ʵ���������Ϊ15%����:
��

��ϰ��ϵ�д�
�����Ŀ
С��ͬѧ��13.9g�����ʵĴ�����Ʒ��̼�������Ȼ��ƵĻ�����90.5gϡ�������ϣ���ַ�Ӧ����÷�Ӧ���������������m���뷴Ӧʱ�䣨t�����������±���ʾ��
������ĿҪ�ش��������⣺
��1��̼������ȫ��Ӧ������CO2������Ϊ g
��2���������������ͼ�У�������Ӧʱ���������������m����ʱ�䣨t���仯�����ߣ�
��3������ȫ��Ӧ��������Һ�����ʵ�������������Na2CO3+2HCl=2NaCl+CO2��+H2O����
| ��Ӧʱ��t/s | t0 | t1 | t2 | t3 | t4 | t5 | t6 |
| ��������m/g | 0 | 0.88 | 1.76 | 2.64 | 3.52 | 4.4 | 4.4 |
��1��̼������ȫ��Ӧ������CO2������Ϊ
��2���������������ͼ�У�������Ӧʱ���������������m����ʱ�䣨t���仯�����ߣ�

��3������ȫ��Ӧ��������Һ�����ʵ�������������Na2CO3+2HCl=2NaCl+CO2��+H2O����
С��ͬѧ��13.9g�����ʵĵĴ�����Ʒ��̼�������Ȼ��ƵĻ�����90.5gϡ�������ϣ���ַ�Ӧ����÷�Ӧ���������������m���뷴Ӧʱ�䣨t�����������±���ʾ��
| ��Ӧʱ��t/s | t0 | t1 | t2 | t3 | t4 | t5 | t6 |
| ��������m/g | 0 | 0.88 | 1.76 | 2.64 | 3.52 | 4.4 | 4.4 |
������ĿҪ�ش��������⣺
��1��̼������ȫ��Ӧ������CO2������Ϊ g
��2���������������ͼ�У�������Ӧ�����������������m����ʱ�䣨t���仯�����ߡ�
��3������ȫ��Ӧ��������Һ�����ʵ�������������Na2CO3��2HCl��2NaCl��CO2����H2O����