��Ŀ����
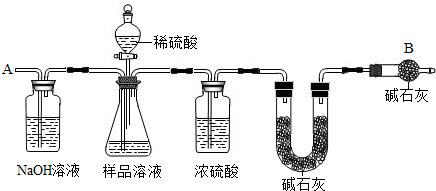
��֪ij������Ʒ�к���NaCl���ʣ�Ϊ�ⶨ��Ʒ�д��������������������ͼ�е�װ�ý���ʵ��(��ܰ��ʾ����ʯ�ҵ���Ҫ�ɷ���NaOH��CaO)����Ҫʵ�鲽�����£�
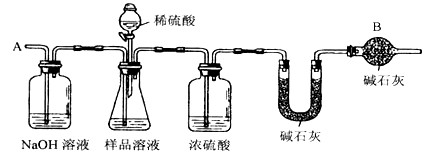
�ٰ�ͼ��װ�����������װ�õ�������
�ڽ�ag��Ʒ������ƿ�У�����������ˮ�ܽ⣬�õ���Ʒ��Һ
�۳���ʢ�м�ʯ�ҵ�U�ܵ��������õ�bg
�ܴӷ�Һ©������ϡ���ᵽ���ٲ�������Ϊֹ
�ݴӵ���A����������һ�����Ŀ���
���ٴγ���ʢ�м�ʯ�ҵ�U�ܵ��������õ�cg
���ظ�����ݺ͢IJ�����ֱ��ʢ�м�ʯ�ҵ�U�ܵ������������䣬Ϊdg
��ش��������⣺
(1)����������ƽ������Ʒʱ�������ƽ��ָ������ƫת��˵��_________________��
(2)װ����NaOH��Һ��������___________________��
(3)װ����Ũ�����������_____________________��
(4)�������Һ©���е�ϡ���ỻ��Ũ����ͬ�����ᣬ�ⶨ�Ľ����_______(��ƫ�ߡ� ƫ�ͻ�)��
(5)����ݵ�Ŀ����________________________��
(6)����Ʒ�д�������������ļ���ʽΪ__________________��
�ڽ�ag��Ʒ������ƿ�У�����������ˮ�ܽ⣬�õ���Ʒ��Һ
�۳���ʢ�м�ʯ�ҵ�U�ܵ��������õ�bg
�ܴӷ�Һ©������ϡ���ᵽ���ٲ�������Ϊֹ
�ݴӵ���A����������һ�����Ŀ���
���ٴγ���ʢ�м�ʯ�ҵ�U�ܵ��������õ�cg
���ظ�����ݺ͢IJ�����ֱ��ʢ�м�ʯ�ҵ�U�ܵ������������䣬Ϊdg
��ش��������⣺
(1)����������ƽ������Ʒʱ�������ƽ��ָ������ƫת��˵��_________________��
(2)װ����NaOH��Һ��������___________________��
(3)װ����Ũ�����������_____________________��
(4)�������Һ©���е�ϡ���ỻ��Ũ����ͬ�����ᣬ�ⶨ�Ľ����_______(��ƫ�ߡ� ƫ�ͻ�)��
(5)����ݵ�Ŀ����________________________��
(6)����Ʒ�д�������������ļ���ʽΪ__________________��
(1)��Ʒ��������
(2)��ȥ�����еĶ�����̼
(3)��ȥ������̼�е�ˮ����
(4)ƫ��
(5)ʹ���ɵĶ�����̼���御���ܱ�U���еļ�ʯ������
(6)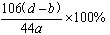
(2)��ȥ�����еĶ�����̼
(3)��ȥ������̼�е�ˮ����
(4)ƫ��
(5)ʹ���ɵĶ�����̼���御���ܱ�U���еļ�ʯ������
(6)

��ϰ��ϵ�д�
 ȫ�ܲ��һ���þ�ϵ�д�
ȫ�ܲ��һ���þ�ϵ�д� ��ɢ˼ά�¿���ϵ�д�
��ɢ˼ά�¿���ϵ�д�
�����Ŀ